A Multicentre Study of Thromboprophylaxis in Pregnancy.
MP Crowley3, C Noone1, JR Higgins2, S O Shea1
1Department of Haematology, Cork University Hospital, Cork, Ireland.
2Anu Research Centre, UCC Department of Obstetrics and Gynaecology, Cork Ireland.
3Centre for Haemostasis and Thrombosis, 1st floor, North Wing, St Thomas' Hospital, Westminster
Abstract
Venous thromboembolism (VTE) is a leading cause of maternal mortality. The risk increases with increasing maternal age, mode of delivery and medical co-morbidities. Thromboprophylaxis with low molecular weight heparin (LMWH) has been shown to be both safe and efficacious. The aim of this study was to prospectively investigate the incidence of maternal risk factors in pregnant women admitted to hospital, to calculate their VTE risk status and to investigate if they were receiving appropriate thromboprophylaxis. All patients admitted to the participating hospitals on the day of investigation were assessed for risk of VTE on the basis of hospital chart review. Five Hundred and forty women were recruited from 16 hospitals. Almost 32% (31.7%) were receiving thromboprophylaxis with LMWH. Just under 80% of patients were on the correct thromboprophylaxis strategy as defined by the RCOG guideline but 49% were under-dosed. The odds of receiving appropriate thromboprophylaxis were significantly increased if the woman was >35 years 0or with parity>3.
Introduction
Venous thromboembolism (VTE) remains a major cause of maternal morbidity and mortality complicating one in a thousand pregnancies. In Ireland, VTE is the leading cause of direct maternal deaths. Recent evidence suggests that the introduction of widespread thromboprophylaxis may have led to a decrease in deaths from VTE. The likelihood of VTE in pregnancy is higher when additional risk factors such as a history of VTE, obesity, increased maternal age and smoking are present. The incidence of these co-morbid conditions is increasing. The potential for a multiplicative effect on overall VTE risk has been shown to be an important factor to consider when risk-stratifying pregnant women. Most risk factors appear to have a greater effect on the incidence of VTE in the postpartum period rather than in the antepartum period. Low molecular weight heparin (LMWH) has been shown to be safe and efficacious in pregnancy. When compared with unfractionated heparin, it has been shown to be associated with a lower risk of osteoporosis and heparin induced thrombocytopenia. It is recommended that a VTE risk assessment is carried out on all pregnant women and thromboprophylaxis is recommended for high risk women. However, despite thromboprophylaxis guidelines, implementation is often variable. The aim of this study was to prospectively investigate the incidence of maternal risk factors in women admitted to maternity hospitals in Ireland, to calculate their VTE risk status and to investigate if they were appropriately receiving thromboprophylaxis.
Methods
All maternity units in the Republic of Ireland were eligible for inclusion. All 20 maternity units were contacted and 16 agreed to participate in the study. Ethical approval was obtained from participating institutions. Any inpatient wards with obstetric patients were included. Labour wards, outpatient clinics, theatres, radiology, and emergency rooms were excluded. Data were collected from a review of hospital charts on standard case report forms by two of the authors (physicians). Each hospital was assessed on one pre-specified day to allow complete and systemic processing of patient data. For inclusion, each patient had to be an inpatient in an eligible ward on the day it was surveyed and be pregnant or have delivered during the current admission. Data abstraction from all eligible charts was completed in selected wards at each hospital on the day of hospital visitation and included patient demographics, admission diagnosis, details of pregnancy, risk factors associated with VTE (as defined in the RCOG Green Top guideline No. 37a12) background medical issues, delivery details (if post-natal) and details of thromboprophylaxis. The study period was September 2011 to November 2012. Patients were excluded from the study if their charts were unavailable for review. Enrolled patients were assessed for risk of VTE and need for thromboprophylaxis in accordance with the RCOG guideline. When assessing whether prophylaxis was compliant with the guideline, both the type and dose of thromboprophylaxis was considered. Duration of dose was recorded if recorded in the medical chart.
Quantitative data was described using mean and standard deviation (SD) if normally distributed and median and interquartile range (IQR) if non-normally distributed. Categorical data was summerised into number (N) and percentage (%). Comparison between groups was performed using Fisher’s exact test for categorical variables. The likelihood of receiving appropriate thromboprophylaxis was calculated by multivariate binary logistic regression analyses. Likelihoods were expressed as odds ratios (ORs). Analysis were performed using Predictive Analytical Software (PASW) Statistics, Version 18.0 using a two-sided type 1 error rate of 0.05.
Results
Baseline characteristics
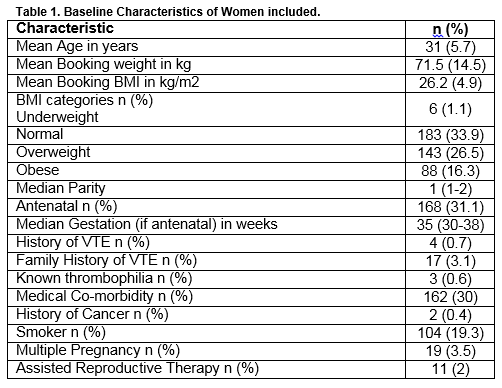
Five hundred and forty women were recruited from 16 hospitals. The mean age at booking was 31(5.7) years (Table 1). One hundred and twenty-four women (23%) were aged over thirty-five at booking. Both antenatal and postnatal women were included; 31.1%(168) of participants were antenatal. The median gestation of antenatal women was 35 (30-38) weeks Eighty-six percent (465) of women had booking weights documented. The mean (SD) booking-weight was 71.5 (14.5) kg. Seventy-eight percent (420) of women had a booking Body Mass Index (BMI) calculated (or a height and weight documented so that a BMI could be calculated). The mean booking BMI was 26.2 (4.9) kg/m2. While 88 (16.3%) women were considered to be obese at booking, it’s important to note that 23 (4.3%) had a BMI>35kg/m2 and 6 (1.1%) had a BMI>40kg/m2. The median parity was 1 with 21.7% of woman having a parity of 3 or more. Four (0.7%) women had a personal history of VTE and 17 (3.1%) had a family history of VTE. Only 7 women had undergone thrombophillia testing; two were heterozgous for the Factor V Leiden mutation and one had antithrombin deficiency. Thirty percent (162) had an underlying medical condition that was unrelated to the pregnancy. A wide variety of conditions were noted. Asthma was the most commonly documented, affecting 5.7% (31); endocrine disorders were the next most common with 3.9% (21) having hypothyroidism, 2% (11) having hyperthyroidism and 3.5% (19) having impaired glucose tolerance or diabetes mellitus. Two women had a history of cancer (breast), three had a history of intravenous drug abuse and one had nephrotic syndrome. Eleven women had varicose veins. Almost one in five women were actively smoking at some stage during the pregnancy. Nineteen of the pregnancies (3.5%) were multiple (all twin) and eleven (2%) were the result of assisted reproductive therapies. No woman had undertaken long-haul travel during her pregnancy.
Pregnancy morbidity
Twenty-eight women (5.2%) had experienced hyperemesis gravidarum and six (1.6%) experienced systemic infection necessitating admission during their pregnancy. Twenty-two (5.9%) had pre-eclampsia/suspected pre-eclampsia. Four women were dehydrated and ten were immobile during their pregnancy.
Delivery
Thirty-three (8.9%) women had prolonged labours and fourteen (3.8%) required midcavity instrumentation. Caesarean sections accounted for 45.7% of deliveries. Twelve (3.2%) women had documented excessive blood loss.
Thromboprophylaxis
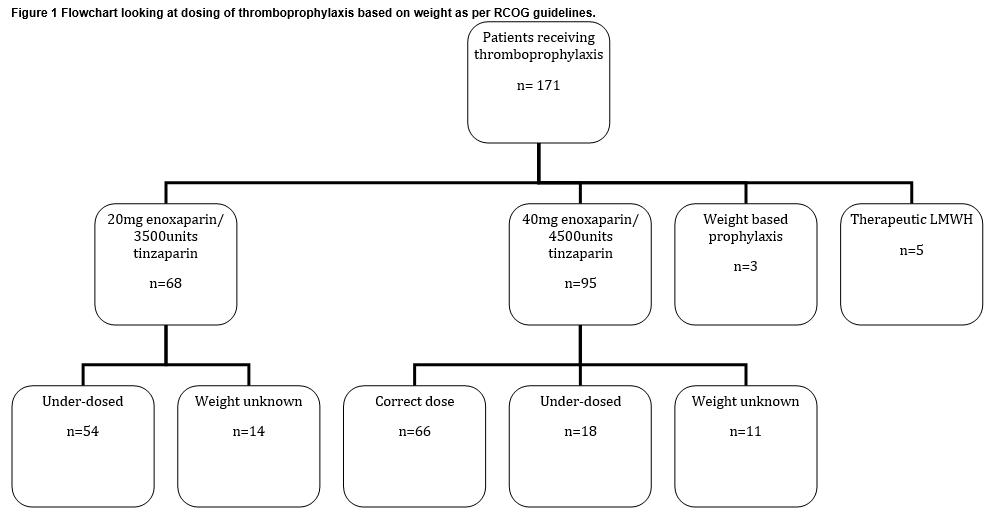
One hundred and seventy-one women (31.7%) were recieving thrombopropylaxis (Figure 1) with LMWH (tinzaparin 92.4%; enoxaparin 7.6%). Most of the thromboprophylaxis was in the postnatal setting: 8.3% of antenatal women; 42.2% of postnatal women. Most (62.6%) of the women on thromboprophylaxis did not have a plan documented in their chart regarding indication or duration of thromboprophylaxis. Of those that did, 44 were having it for one week postpartum, 1 was having it for six weeks postpartum, 5 were having it both antenatally and for six weeks postpartum and 5 were having it while admitted. Of women recieving enoxaparin, 25% recieved 20mg daily and 75% recieved 40mg daily. Of those recieving tinzaparin, 42.6% recieved 3,500 units daily and 57.3% recieved 4,500 units. Four women received therapeutic tinzaparin and three received weight-adjusted prophylactic doses. Of those women with weights documented, 49% were under-dosed, based on their booking weight
Risk assessments
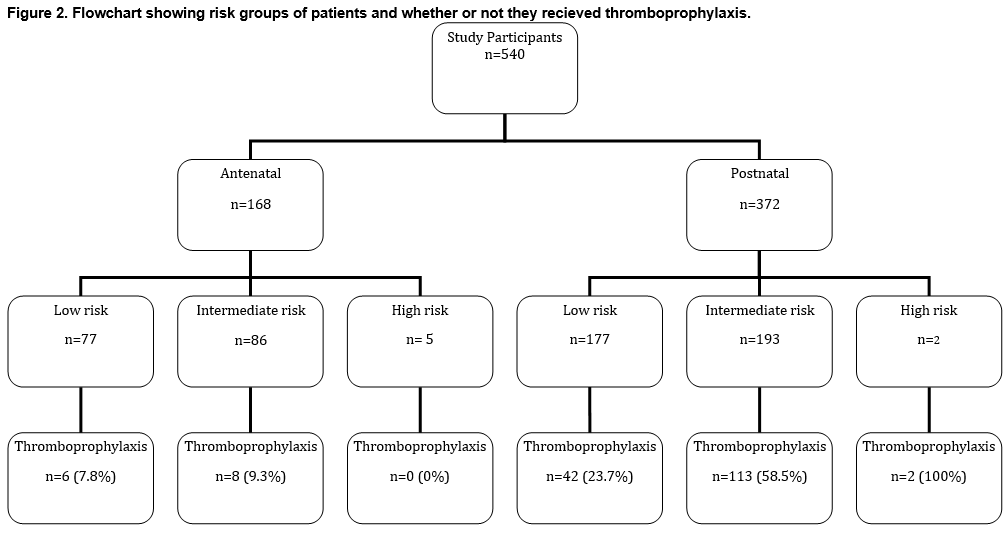
The investigators carried out a VTE risk assessment (according RCOG guidelines) on all women, based on documented risk factors (Figure 2). In the antenatal group, women were considered to be high risk if they had either a history of a single previous VTE associated with a thrombophilia/a family history of VTE/unprovoked VTE/oestrogen related VTE or had a history of recurrent VTE (>1). It is advised that these women should be on antenatal thromboprophylaxis. Five(3%) women were considered high risk. Women were considered intermediate risk if they had a previous VTE that did not meet high risk criteria, known thrombophillia and no history of VTE, a medical co-morbitiy as outlined in the guideline, a surgical procedure or two or more risk factors as outlined in the guideline (all were inpatients). Antenatal thromboprophylaxis should be considered in these women. Eighty-six (51.2%) women were considered intermediate risk. Women were considered low risk if they did not meet the criteria for high or intermediate risks. Mobilisation and avoidance of dehydration is recommended in this group. Seventy-seven (45.8%) women were considered low risk.
In the postnatal group, women were considered high risk if they had ever had a VTE or had an indication for antenatal thromboprophylaxis. It is advised that they recieve at least 6 weeks of postnatal thromboprophylaxis. Two (0.5%) women were considered high risk. Women were considered intermediate risk if they had a caesarean section in labour, they had an asymptomatic thrombophilia, BMI>40kg/m2, their hospital admission was prolonged, they had a medical co-morbidity as defined in the guideline or had two or more risk factors as defined by the guideline. It was advised that these women recieve at least 7 days thromboprophylaxis and that extended thromboprophylaxis should be considered for women with 3 or more risk factors. One hundred and ninety-three women (51.9%) were considered intermediate risk. Women were considered low risk if they didn’t meet criteria for other risk groups. Again, mobilisation and avoidance of dehydration was recommended for these women. One hundred and seventy-seven women (47.6%) were considered low risk. Overall, 48 (18.9%) low risk women were on thromboprophylaxis, 121 (43.4%) intermediate risk women were on thromboprophylaxis and 2 (28.6%) high risk women were on thromboprophylaxis.
Different types of maternity units were involved in this study including large obstetric units with formal obstetric haematology services, stand alone maternity hospitals and smaller obstetric units attached to general hospitals. When centres with formal obstetrichaematology services were compared to other units, it was found that they were similar in terms of patient risk profiles (p=0.164) and in terms of thromboprophylaxis use (p=0.248). Overall, 73.9% of patients were on the correct thromboprophylaxis strategy as defined by the RCOG guideline. The odds of receiving appropriate thromboprophylaxis were significantly increased if the woman was aged 35 or over (OR 2.311; 95%CI 1.415-3.775;p=0.001) or if parity was greater than 3 (OR1.775; 95%CI1.078-2.923;p=0.024) but not if BMI >30 (p=0.827) orpatients had a history of VTE (p=0.999).
Discussion
Twenty-six percent of patients in this study were not on an appropriate thromboprophylactic regimen, according to RCOG guidelines. This included 5 antenatal women who were considered high risk and 80 postnatal women who were considered intermediate risk but were not on pharmacological thromboprophylaxis. Of those women that received thromboprophylaxis, almost two-thirds had no documented thromboprophylaxis plan in their notes. Obstetric units are busy with a high turnover of pregnant women so if thromboprophylaxis is commenced, a plan regarding drug, dose and duration should be documented to ensure that women recieve optimal care. Almost half the women who recieved thromboprophylaxis were on too low a dose. A wide variety of obstetric units were looked at in this study (range of inpatients per unit: 11-105) which reflects current obstetric practice in Ireland. The mean age at booking was 31 with almost a quarter of women aged over 35. Women aged over 35 are at an increased risk of pregnancy related morbidity17,18. It is a concerning trend that they are forming a large proportion of obstetric cohorts given their high-risk nature. The mean BMI was 26.2kg/m2 with 55% of patients being overweight or obese at booking. Worryingly, 22% of patients did not have a BMI (or height and weight) documented at booking. Increased BMI is associated with pregnancy and neonatal morbidity19. It should be identified as an issue as early as possible so that remedial action can be instigated. In the context of this study, as BMI forms part of both the antenatal and postnatal risk assessment, patients may have been considered low risk when in fact they were intermediate risk and may explain why some low risk patients received thromboprophylaxis.
Few patients had a personal or family history of VTE. Almost one third had an underlying medical condition which could impact the pregnancy. Almost one in five women were actively smoking at some stage during the pregnancy. This is despite the fact that it has long been recognised as being harmful in pregnancy20. Moreover, there have been several campaigns, both local and national, highlighting the dangers of smoking in pregnancy and resources dedicated to smoking cessation21. Since this study, a clinical practice guideline for venous thromboprophylaxis in pregnancy22 has been published by the Institute of Obstetricians and Gynaecologists, Royal College of Physicians of Ireland and the Irish Haematology Society in 2013. It is hoped that this will lead to a simplified risk assessment of pregnant women and improve appropriate use of thromboprophylaxis. Following implementation of this guideline, we plan to repeat this study.
Conflict of interest
MP Crowley and C Noone received unrestricted educational grants from Leo Pharmaceuticals.
Correspondence:
Maeve P Crowley, Centre for haemostasis and thrombosis, 1st floor, North Wing, St, Thomas' Hospital, Westminster Bridge Road, London SE1 7EH
Email address: [email protected]
References
1. Chang J, Elam-Evans LD, Berg CJ, Herndon J, Flowers L, Seed KA, Syverson CJ. Pregnancy-related mortality surveillance--United States, 1991--1999. MMWR Surveill Summ. 2003 Feb 21;52:1-8.
2. Confidential Maternal Death Enquiry in Ireland, Report for Triennium 2009-2011, Cork: MDE, August 2012
3. Centre for Maternal and Child Enquiries (CMACE). Saving Mothers’ Lives: reviewing maternal deaths to make motherhood safer: 2006–08. The Eighth Report on Confidential Enquiries into Maternal Deaths in the United Kingdom. BJOG 2011;118(Suppl. 1):1–203.
4. Jacobsen AF, Skjeldestad FE, Sandset PM. Incidence and risk patterns of venous thromboembolism in pregnancy and puerperium--a register-based case-control study. Am J Obstet Gynecol. 2008 Feb;198:233.e1-7. Epub 2007 Nov 12.
5. James AH, Tapson VF, Goldhaber SZ. Thrombosis during pregnancy and the postpartum period. Am J Obstet Gynecol. 2005 Jul;193:216-9.
6. Larsen TB, Sørensen HT, Gislum M, Johnsen SP. Maternal smoking, obesity, and risk of venous thromboembolism during pregnancy and the puerperium: a population-based nested case-control study. Thromb Res. 2007;120:505-9. Epub 2007 Jan 25.
7. Ghaji N, Boulet SL, Tepper N, Hooper WC.Trends in venous thromboembolism among pregnancy-related hospitalizations, United States, 1994-2009. Am JObstet Gynecol 2013;209:433.e1-8.
8. Kevane B, Donnelly J, D'Alton M, Cooley S, Preston RJ, Ní Ainle F. Risk factors for pregnancy-associated venous thromboembolism: a review. J Perinat Med. 2014 Jul;42:417-25.
9. Sultan AA, Tata LJ, West J, Fiaschi L, Fleming KM, Nelson-Piercy C, Grainge MJ. Risk factors for first venous thromboembolism around pregnancy: a population-based cohort study from the United Kingdom. Blood. 2013;121:3953-3961.
10. Greer IA, Nelson-Piercy C. Low-molecular-weight heparins for thromboprophylaxis and treatment of venous thromboembolism in pregnancy: a systematic review of safety and efficacy. Blood. 2005 Jul 15;106:401-7. Epub 2005 Apr 5. Review.
11. Nelson-Piercy C, Powrie R, Borg JY, Rodger M, Talbot DJ, Stinson J, Greer IA. Tinzaparin use in pregnancy: an international, retrospective study of the safety and efficacy profile. Eur J Obstet Gynecol Reprod Biol. 2011 Dec;159:293-9.
12. Nelson-Piercy C, MacCallum P, Mackillop L. RCOG Green Top Guideline No. 37a Reducing the risk of thrombosis and embolism during pregnancy and the puerperium. London, United Kingdom:RCOG;2009.
13. Yu HT, Dylan ML, Lin J, Dubois RW. Hospitals' compliance with prophylaxis guidelines for venous thromboembolism. American journal of health-system pharmacy : AJHP : official journal of the American Society of Health-System Pharmacists. 2007;64:69-76. Epub 2006/12/26.
14. Cohen AT, Tapson VF, Bergmann JF, Goldhaber SZ, Kakkar AK, Deslandes B, et al. Venous thromboembolism risk and prophylaxis in the acute hospital care setting (ENDORSE study): a multinational cross-sectional study. Lancet. 2008;371(9610):387-94. Epub 2008/02/05.
15. Rashid ST, Thursz MR, Razvi NA, Voller R, Orchard T, Shlebak AA. Venous thromboprophylaxis in UK medical inpatients. Journal of the Royal Society of Medicine. 2005;98:507-12. Epub 2005/11/02.
16. Cregan A, Higgins JR, O’Shea S. Implementation of thromboprophylaxis guidelines. Ir Med J. 2013; Mar,106 :80-82.
17. Jolly M, Sebire N, Harris J, Robinson S, Regan L. The risks associated with pregnancy in women aged 35 years or older. Hum Reprod. 2000 Nov;15(11):2433-7.
18. Kenny LC, Lavender T, McNamee R, O'Neill SM, Mills T, Khashan AS. Advanced maternal age and adverse pregnancy outcome: evidence from a large contemporary cohort. PLoS One. 2013;8:e56583. doi: 10.1371/journal.pone.0056583. Epub 2013 Feb 20.
19. Avcı ME, Sanlıkan F, Celik M, Avcı A, Kocaer M, Göçmen A. Effects of Maternal Obesity on Antenatal, Perinatal, and Neonatal Outcomes. J Matern Fetal Neonatal Med. 2014 Oct 20:1-13. [Epub ahead of print]
20. Lowe CR. Effect of mothers' smoking habits on birth weight of their children.Br Med J. 1959 Oct 10;2:673-6.
21. Health Service Executive. Quit. http://www.giveupsmoking.ie
22. Institute of Obstetricians and Gynaecologists Royal College of Physicians of Ireland, HSE Clinical Care Programme in Obstetrics and Gynaecology, Irish Haematology Society. Clinical Practice Guideline: Venous Thromboprophylaxis in Pregnancy. Version 1.0; Guideline No. 20. Nov 2013. https://www.rcpi.ie/content/docs/000001/1392_5_media.pdf
(P567)