Epithelioid Angiomyolipoma – a case report and review of the literature
F Keane1, M Greally1, E Harrold1, C Barrett2, J McCaffrey1
1Department of Medical Oncology, Mater Misericordiae Hospital, Dublin
2Department of Pathology, Mater Misericordiae Hospital, Dublin.
Abstract
Herein we present the case of a 43-year-old female in whom a left renal mass was identified incidentally on imaging performed for staging of a newly diagnosed breast carcinoma. The mass was resected and histologic examination and immunohistochemistry confirmed a diagnosis of epithelioid angiomyolipoma.
Introduction
This case highlights the uncommon diagnosis of epithelioid angiomyolipoma(EAML), incidentally diagnosed in a 43 year-old female at our institution. We present the diagnostic and management issues associated with this unusual diagnosis.
Case Report
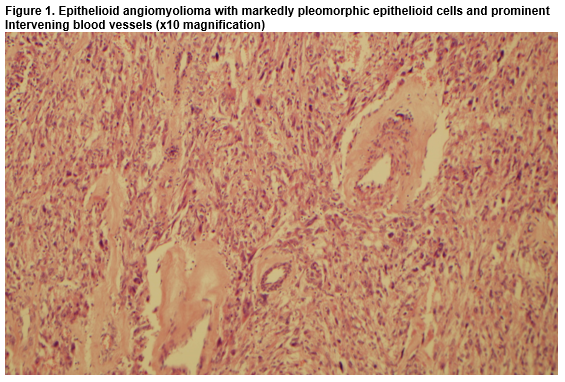
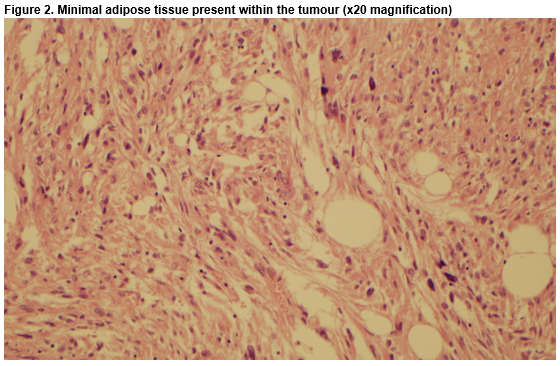
A 43-year-old female was referred to the oncology service following a diagnosis of a pT3N1a invasive ductal carcinoma of the left breast, which was HER-2 positive, hormone receptor negative. During her work-up a whole-body staging Positron Emission Tomography/Computed Tomography (PET/CT) scan revealed a 2.8cm mass, exophytic to the lower pole of the left kidney, associated with low/intermediate grade metabolic activity. Radiological appearances were initially deemed suspicious for renal cell carcinoma (RCC). To investigate further, a Triphasic Renal CT was performed, which confirmed a left renal mass, measuring 4.2cm x 3.0cm. The patient was asymptomatic, physical examination was unremarkable and urinalysis was negative for haematuria. She had no personal or family history of Tuberous Sclerosis Complex (TSC). Following multidisciplinary discussion between Medical Oncology, Urology, Pathology and Radiology specialists a consensus decision was reached to surgically resect the renal mass in the brief time interval between the patient undergoing breast surgery and commencement of adjuvant systemic treatment for her cancer. The patient elected to have a bilateral mastectomy upfront, and subsequently a partial nephrectomy was performed. Histology and immunohistochemistry revealed a malignant neoplasm consistent with the rather unexpected diagnosis of epithelioid angiomyolipoma (Fig.1, Fig. 2).
At follow-up multidisciplinary discussion, a management plan of surveillance imaging and regular clinical review was agreed upon. Of note, this patient’s mother was diagnosed with metastatic breast cancer at age twenty-nine. No genetic predisposition, including BRCA mutation, has been identified. The patient is now over twelve months post bilateral mastectomy and partial nephrectomy. She has completed adjuvant Doxorubicin/Cyclophosphamide/Paclitaxel(AC-T) chemotherapy and twelve months of adjuvant Trastuzumab, with no evidence of disease recurrence to date.
Discussion
With recent improvements in the sensitivity of Computed Tomography(CT) imaging, an increase in the incidence of incidental renal lesions has been observed1. However, it is now established that renal tumours measuring 3cm or less are unlikely to grow quickly and metastasise2. The management of such lesions is an important consideration in patients receiving regular imaging, such as oncology patients. The management dilemma regarding whether these patients should have surgical resection of renal tumours before or after completion of their oncology treatment, is becoming more common in this patient cohort. Renal epithelioid angiomyolipoma was first described in 1994, as a variant of the commonly described benign renal angiomyolipoma(AML). Since then it has become an increasingly recognised diagnosis, and was first included in the WHO Classification of Renal Tumours in 2004. EAMLs account for 4.6% of all AMLs. Median age of diagnosis is 49.7 years. A female preponderance has been reported3.
Unlike classical AMLs, EAMLs are associated with significant malignant potential, with metastatic disease present in up to 30% of cases at diagnosis4. Due to the small number of reported cases, criteria have not been established to predict which EAMLs are more likely to have malignant potential. However, a number of features are thought to be predictive when present, including associated TSC, increasing tumour size, extra-renal extension, cellular atypia and mitotic activity5,6. The prevalence of EAMLs is less than 1 in 10,0007. Approximately 20% of EAMLs occur in subjects with TSC, but may also occur sporadically7,8. Radiologically, classical AMLs are typically easily distinguishable from other renal lesions on CT, due to their characteristic intra-tumoral fat content. However, EAMLs are less easily identifiable due to elevated cellular content and paucity of intratumoral fat, rendering them difficult to differentiate radiologically from other renal masses, such as RCC9.
EAMLs are distinct histologically due to the predominance of epithelial cells on histological examination. Other characteristic features include cellular pleomorphism, abundant eosinophilic cytoplasm, coagulative tumour necrosis, mitotic figures and atypical mitoses3. Immunohistochemical positivity for HMB-45 and negativity for S100 and cytokeratins distinguish the diagnosis from other differentials, including RCC and melanoma. Although surgery is the cornerstone of management in EAMLs, numerous case studies have reported disease recurrence despite tumour resection3,4,6,9. There is no standard systemic therapy for EAML. Various chemotherapy regimens have shown little activity in anecdotal reports. Mammalian target of rapamycin (mTOR) inhibitors have demonstrated therapeutic potential in small studies, but prospective data is lacking10.
Conflicts of Interest
The authors have no conflicts of interest to disclose.
Correspondence: Dr. Fergus Keane, Department of Medical Oncology, Level 7, Whitty Building, Mater Misericordiae Hospital, Dublin.
Email: [email protected]
References
1. O’Connor SD, Pickhardt PJ, Kim DH, Oliva MR, Silverman SG. Incidental finding of renal masses at Unenhanced CT: Prevalence and analysis of features for guiding management. American Journal of Roentgenology. 2011 Jul;197(1):139–45.
2. Bosniak MA, Birnbaum BA, Krinsky GA, Waisman J. Small renal parenchymal neoplasms: Further observations on growth. Radiology. 1995 Dec;197(3):589–97.
3. He W, Cheville JC, Sadow PM, Gopalan A, Fine SW, Al-Ahmadie HA, Chen Y-B, Oliva E, Russo P, Reuter VE, Tickoo SK. Epithelioid angiomyolipoma of the kidney: Pathological features and clinical outcome in a series of consecutively resected tumors. Modern Pathology. 2013 Apr 19;26(10):1355–64.
4. Brimo F, Robinson B, Guo C, Zhou M, Latour M, Epstein JI. Renal Epithelioid Angiomyolipoma with Atypia: A series of 40 cases with emphasis on Clinicopathologic Prognostic indicators of malignancy. The American Journal of Surgical Pathology. 2010 Apr:1.
5. Belanger EC, Dhamanaskar PK, Mai KT. Epithelioid angiomyolipoma of the kidney mimicking renal sarcoma. Histopathology. 2005 Oct;47(4):433–5.
6. Nese N, Martignoni G, Fletcher CD, Gupta R, Pan C-C, Kim H, Ro JY, Hwang IS, Sato K, Bonetti F, Pea M, Amin MB, Hes O, Svec A, Kida M, Vankalakunti M, Berel D, Rogatko A, Gown AM. Pure Epithelioid PEComas (so-called Epithelioid Angiomyolipoma) of the kidney. The American Journal of Surgical Pathology. 2011 Feb;35(2):161–76.
7. Lopater J, Daniel L, Akiki A, Boissier R, Lechevallier É, Coulange C. Angiomyolipome épithélioïde rénal. Progrès en Urologie. 2009 Jul;19(7):457–61.
8. Lane BR, Aydin H, Danforth TL, Zhou M, Remer EM, Novick AC, Campbell SC. Clinical Correlates of renal Angiomyolipoma subtypes in 209 patients: Classic, fat poor, Tuberous sclerosis associated and Epithelioid. The Journal of Urology. 2008 Sep;180(3):836–43.
9. Lei JH, Liu LR, Wei Q, Song TR, Yang L, Yuan HC, Jiang Y, Xu H, Xiong SH, Han P. A Four-Year follow-up study of renal Epithelioid Angiomyolipoma: A multi-center experience and literature review. Scientific Reports. 2015 May 5;5:10030
10. Wolff N, Kabbani W, Bradley T, Raj G, Watumull L, Brugarolas J. Sirolimus and Temsirolimus for Epithelioid Angiomyolipoma. Journal of Clinical Oncology. 2010 Jan 4;28(5):e65–8.
(P604)