Antimicrobial-resistant faecal organisms in algae products marketed as health supplements
L Ryan1, M. Molloy1, L. Evans1, A. Quinn1, E. Burke1, E. McGrath1, M. Cormican2
1Department of Microbiology, Galway University Hospital, Galway, Ireland
2School of Medicine, National University of Ireland, Galway
Abstract
Dietary supplements are increasingly popular in Irish society. One of these is blue-green algae which is used with a variety health benefits in mind. A batch of Chlorella powder was found to be contaminated with Salmonella species in Ireland in 2015. This prompted additional testing of a total of 8 samples of three different products (Chlorella, Spirulina and Super Greens), for other faecal flora and antimicrobial resistance in any bacteria isolated. All 8 samples cultured enteric flora such as Enterococci, Enterobacteriaceae and Clostridium species. Antimicrobial susceptibility testing revealed one isolate with extended-spectrum β-lactamase (ESBL) activity and one with carbapenemase activity. Clinicians caring for vulnerable patients should be aware of the potential risk of exposure to antimicrobial resistant bacteria associated with these products.
Introduction
Chlorella, Spirulina and Super Greens are three products marketed in Ireland as food supplements, beneficial to health. All three products are comprised in whole of green algae, blue-green or microalgae. The algae that form the basis of these products can be cultivated by a number of methods, including open systems in direct contact with the general environment. Most micro-algal cultivation is in Asia and Australia. China is considered one of the leading producers of Chlorella and Spirulina1,2. In 2015, as part of official control sampling, Salmonella Rissen was identified a sample of Chlorella powder on the market in the Ireland. Implicated batches of the powder were recalled from consumers3. Arising from this incident, we carried out further testing on these products, for a range of faecal flora and to assess antimicrobial resistance of any Enterobacteriaceae or Enterococci isolated.
Methods
Eight samples (5 Chlorella, two Spirulina and one Super Greens) were obtained from retail outlets in one town in Ireland. Twenty-five gram samples were diluted in 225g of peptone buffer solution and cultured for 24 hours at 37°C. A negative control of 250g of peptone buffer solution was cultured in parallel. Broths were sub-cultured to two Columbia blood agar plates (CBA) for aerobic and anaerobic incubation, Mueller-Hinton Agar, and selective chromogenic agars for vancomycin-resistant Enterococci, meticillin-resistant Staphylococcus aureus, extended-spectrum beta-lactamase producing Enterobacteriaceae and carabpenemase-producing Enterobacteriaceae (Biomérieux ChromID™ VRE/MRSA/ESBL Agars, Oxoid Brillance™ CRE Agar) and xylose lysine deoxycholate (XLD Oxoid) agar for Salmonella spp. (OXOID). Plates were incubated at 37°C for a further 24 hours. A representative of each distinct colony type cultured was identified using MALDI-TOF MS (Bruker Maldi Biotyper™). For Enterobacteriaceae and Enterococci, antimicrobial susceptibility testing was performed by disc diffusion by the EUCAST method. Susceptibility to the following antimicrobials was assessed – amoxicillin, vancomycin, linezolid, quinupristin-dalfopristin and gentamicin for Enterococcus spp. and amoxicillin, amoxicillin-clavulanic acid, piperacillin-tazobactam, cefotaxime, ertapenem, meropenem, ciprofloxacin, gentamicin, amikacin, cefpodoxime, cefpodoxime-clavulanic acid and cefoxitin for Enterobacteriacae.
Isolates of Klebsiella spp. were tested for extended-spectrum beta-lactamase (ESBL) production using cefpodoxime (10µg) (CPD10) and cefpodoxime with clavulanate by EUCAST methods and criteria. Enterobacter spp. were assessed for ESBL using cefepime ESBL E-test (cefipime/cefipime-clavulanate or PM/PML) according to manufacturer’s instructions. Enterobacteriaceae identified as ESBL or with reduced susceptibility to meropenem were tested by rtPCR for a range of common ESBL and CPE genes.
Results
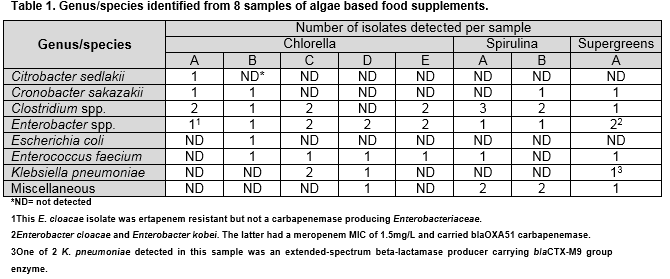
Enteric type flora (Clostridium spp., Enterococcus faecium or Enterobacteriaceae) were recovered from all 8 samples tested as illustrated in Table 1. All Enterococcus spp. were vancomycin susceptible. The K. pneumoniae isolated from the sample of Super Greens carried blaCTX-M Group 9 b-lactamase and among agents tested was susceptible only to amikacin, cefoxitin, ertapenem, meropenem and piperacillin-tazobactam. An isolate of Enterobacter kobei, also from the Super Greens sample, carried blaOXA-51 carbapenemase.
Discussion
Antimicrobial resistance in Enterobacteriaceae found in other foods, especially meat products, is a well reported phenomenon and consumption of contaminated foods is considered to be one mechanism by which humans acquire or become colonised with multidrug-resistant organisms11. This project indicates that these algae based products, promoted as food supplements with claims of health benefits, and often consumed without cooking, are frequently contaminated with faecal type bacteria, which in some instances carry antimicrobial resistance determinants of significant public health concern. Of note half of the samples contain Cronobacter sakazakii which has been associated with outbreaks of serious infection in neonatal intensive care units related to infant formula and in immunocompromised adults4,5. Reported sites of infection include urinary, biliary tract, appendicitis, wound infections and pneumonia5. These products may be unlikely to be given to neonates or infants, however they are not stated to be unsuitable for infants or immunocompromised people. Consumption of these products could lead to gastrointestinal colonisation with C. sakazakii6. Colonisation represents a reservoir with potential for subsequent infection.
There are some previous reports from Germany and the USA of contamination of such products with toxins (microcystins) and material such as rat hairs and insect fragments, and two reports, from Mexico and China, of microbial contamination from 1976 and 1981.7-10 In Ireland the food laws in place relating to food supplements are summarised in Guidance Note 21 from the FSAI and include legislation relating to general food laws, traceability, food hygiene and safety.3 The products included in this study were compliant with the legislation in terms of traceability, food hygiene and safety. There are no microbiological criteria set out specifically in relation to food supplements; they are subject to microbiological criteria as detailed in Guidance Note Three from the FSAI relating to ready-to-eat foods placed on the market3. Enterobacteriaceae and E. coli in ready-to-eat foods at <102 cfu/g and <20 cfu/g, respectively are considered as satisfactory3. Enterobacteriaceae are a general hygiene indicator microorganism. Antimicrobial resistance of Enterobacteriaceae is not generally assessed in relation to microbiological analysis of food and antimicrobial resistance of Enterobacteriaceae has not to date been considered in relation to setting criteria for acceptability.
As this is a small study, it is not possible to conclude that contamination of these products with antimicrobial resistant organisms is present in all available products, however there is sufficient basis for clinicians caring for vulnerable patients, to be aware of the potential risk of exposure to antimicrobial resistant bacteria associated with these products. More extensive surveys are also required to inform consumers, producers and policy makers on the extent to which antimicrobial resistant bacteria are present in ready to eat foods so as to inform decisions regarding the need for improved practices and or regulation in relation to antimicrobial resistant bacteria in food.
Correspondence:
Laura Ryan, Department of Microbiology, Galway University Hospital, Galway, Ireland
Email: [email protected]
Conflict of interest
None to declare.
References:
1. Christien Enzing, Matthias Ploeg, Maria Barbosa, Lolke Sijtsma. Microalgae-based products for the food and feed sector: an outlook for Europe. JRC scientific and policy reports 2014
2. Yung-Ming Lu, Wen-Zhou Xiang, Yong-Huang Wen. Spirulina (Arthorspira) industry in inner Mongolia of China: Current status and prospects. J appl Phycol (2011) 23: 265-9
3. The Food Safety Authority of Ireland. www.fsai.ie Accessed April 2017.
4. Simmons BP, Gefland MS, Haas M, Metts L, Ferguson J. Enterobacter sakazakii infections in neonates associated with intrinsic contamination of a powdered infant formula. Infect Control Hosp Epidemiol. 1989 Sep; 10 (9): 389-401.
5. Holý O. Forsythe S. Cronobacter spp. as emerging causes of healthcare-associated infection. J Hosp Infect. 2014 Mar; 86(3): 169-77.
6. Liu H, Cui JH, Cui ZG, Hu GC, Yang YL, Li J, Shi YW. Cronobacter carriage in neonate and adult intestinal tracts. Biomed Environ Sci. 2013 Oct; 26(10: 861-4.
7. Heussner AH, Mazija L, Fastner J, Dietrich DR. Toxin content and cytotoxicity of algal dietary supplements. Toxicol Appl Pharmacol 2012 Dec 1; 265(2): 263-71.
8. Nakashima MJ, Angold S, Beavin BB, Bradicich RB, Decker SJ, Dzidowski GR, Levesque E, Locatelli RG, Mably M, Paredes A. Extraction of light filth from Spirulina powders and tablets: collaborative study. J Assoc Off Anal Chem 1989 May-Jun; 72(3): 451-3.
9. Gonzalea Arroyo S, Luna Lagunes RM, Hernandex Velarde R, Soriano Lucio P, Torres Gillardo J. Preliminary study of bacterial contamination in a seminatural culture of Spirulina. Salud Publica Mex. 1976 Jul-Aug; 18 (4): 705-10.
10. Wu JF, Pond WG. Amino acid composition and microbial contamination of Spirulina maxima, a blue-green alga, grown on the effluent of different fermented animal wastes. Bull Environ Contam Toxicol. 1981 Aug; 27(2): 151-9.
11. Liu YY, Wang Y, Walsh TR, Yi LX, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, Yu LF, Gu D, Ren H, Chen X, Lv L, He D, Shou H, Liang Z, Liu JH, Shen J. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 2016 Feb; 16(2): 161-8.
(P624)