Seasonal Variation in the Emergency Department Prevalence Of Sepsis
McNevin C1, McDowell R2, Ni Shearcaigh A3, Wakai A3,4
1Beaumont Hospital, Dublin 9, Ireland.
2RCSI Department Of General Practice, Division Of Population Health Sciences (Phs), Dublin 2, Ireland.
3 Department Of Emergency Medicine, Beaumont Hospital, Dublin 9, Ireland.
4 Emergency Care Research Unit (ECRU), RCSI Department Of General Practice, 123 St Stephens Green, Dublin 2, Ireland.
Abstract
The incidence and mortality of sepsis and severe sepsis in hospitalised patients is seasonal and consistently highest during the winter. The primary aim of this study was to measure the seasonal variation in the prevalence of emergency department (ED) patients with sepsis. This cross-sectional study was performed over two four-week periods in the summer and in the winter, respectively. The clinical records of all patients presenting to the ED during the study periods were retrospectively screened to determine if they met the criteria for “uncomplicated” sepsis and severe sepsis or septic shock. The prevalence of “uncomplicated” sepsis was higher in the winter (43.9 per 1000) compared to the summer (30.7 per 1000). The prevalence of severe sepsis or septic shock was also higher in the winter (17.7 per 1000) compared to the summer (11.7 per 1000). This quantitatively demonstrates the increased ED burden of sepsis in the winter that can be used to inform healthcare planning and resource allocation.
Introduction
In Ireland, up to 60% of all hospital deaths have a sepsis or infection diagnosis1. Due to the fact that the majority of patients with severe sepsis are admitted to intensive care units from the emergency department (ED)2, knowledge of seasonal variations in the incidence of sepsis and severe sepsis has important implications for critical care resources and for the allocation of limited healthcare resources3. Meanwhile, the incidence and mortality of sepsis and severe sepsis have been shown to be seasonal and consistently highest in during the winter3. Although we have previously measured the prevalence of ED patients who meet the criteria for “uncomplicated” sepsis in a summer month4, there is no published study reporting the seasonal variation in the epidemiology of sepsis in the Irish ED setting. The primary aim of this study was to measure the seasonal variation in the prevalence of patients who meet the criteria for sepsis in an Irish ED.
Methods
This study was performed in the ED of Beaumont Hospital, Dublin. The annual census of the ED is approximately 50,000. The Beaumont Hospital Ethics (Medical Research) Committee approved the study. Data from two one-month periods (July to August 2015 and February 2016, respectively) was analysed to identify patients who met the criteria for “uncomplicated” sepsis and “severe sepsis or septic shock” using the Health Service Executive (HSE) Sepsis Screening Form4,5. The clinical records were retrospectively reviewed within 24 hours of ED arrival. Data was analysed with descriptive statistics and reported as frequencies, proportions, percentages, medians and interquartile ranges (IQR). Point estimates (p-values) and interval estimates (95% confidence intervals [CIs]) for the proportion of patients who met the criteria for “uncomplicated” sepsis and for severe sepsis or septic shock were calculated. The outcome sepsis classification/status was treated as an ordered categorical variable; a generalised ordered logistic model6 was used to model sepsis status as a function of season, adjusting for gender and age. It was hypothesised that the effects associated with age on sepsis status could vary by season and hence interactions between season and age were retained in the model. Violations of the proportionality assumption were considered not just of statistical significance but of clinical importance. Age was centred around the median value (47 years). Any relevant odds ratio (OR) included the 95% CI. A p-value of less than 0.05 was considered statistically significant. Analyses were performed using Microsoft Excel 2010, Stata 13.0 SE, the gologit2 module for Stata and SPSS version 21.0.
Results
Patient characteristics
Overall, 3,585 patients attended the ED during the summer month and 3,784 patients attended during the winter month (Table 1). The median age of attendees was slightly higher in the winter (median = 48.0 years; IQR: 31.0 – 68.0 years) than in the summer (median = 46.0 years; IQR: 31.0 – 66.0 years) although the age distributions of the ED patients did not differ significantly between the two months (p=0.20).

Prevalence of sepsis
The distribution of ED patients according to sepsis category varied between the two seasons (p=0.001), with a greater proportion of patients classified as having either “uncomplicated” sepsis or severe sepsis/septic shock in the winter month than in the summer month (Table 1). The proportion of ED patients who met the criteria for “uncomplicated” sepsis was 3.07% (95% CI 2.49 to 3.65%) in the summer month and 4.39% (95% CI 3.72 to 5.05%) in the winter month. The proportion of ED patients who met the criteria for severe sepsis or septic shock was 1.17% (95% CI 0.81 to 1.54%) in the summer month and 1.77% (95% CI 1.34 to 2.2%) in the winter month.
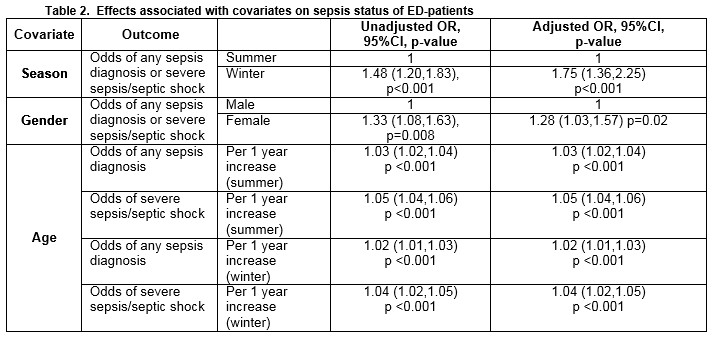
Effect of season, gender and age
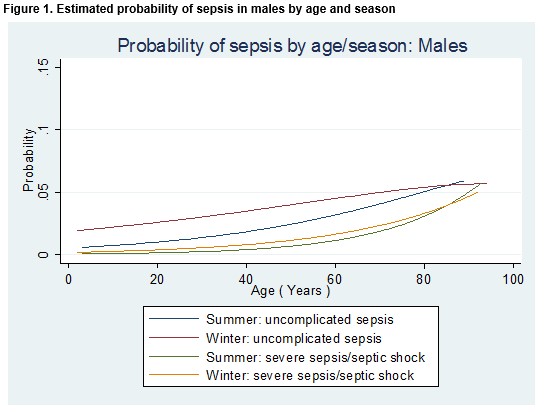
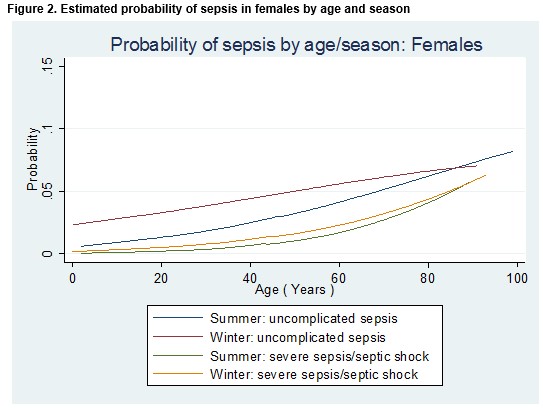
The effect of season, gender and age on the odds of a diagnosis of any type of sepsis or a diagnosis of severe sepsis/septic shock is detailed in Table 2. The odds of any sepsis diagnosis or a diagnosis of severe sepsis/septic shock was 48% higher in the winter than in the summer month (OR = 1.48; 95% CI 1.20 to 1.83; p<0.001). After controlling for age and gender (i.e. for a male patient of median age (47 years), the odds of any sepsis diagnosis or a diagnosis of severe sepsis/septic shock was 75% higher in the winter than in the summer month (OR = 1.75; 95% CI 1.36 to 2.25; p<0.001). Among patients of the same age and who attended the ED in the same month, females were 28% more likely than males to receive a diagnosis of any type of sepsis diagnosis or a diagnosis of severe sepsis/septic shock (OR = 1.28; 95% CI 1.03 to 1.57; p=0.02). Meanwhile, the probability of a diagnosis of either “uncomplicated” sepsis or severe sepsis/septic shock was higher in the winter than in the summer and increased with age, but for older patients the differential between the seasons was diminished (Figures 1 and 2).
Source of sepsis
The respiratory system was the most common source of sepsis in both the summer month (n=56; 36.8%) and the winter month (n= 107; 45.9%), although the difference between seasons in these proportions was not statistically significant (p=0.07). Among patients for whom the respiratory system was the source of sepsis, the odds of having severe sepsis/septic shock was 12.0% higher in the winter month than the summer month, but this difference was not statistically significant (OR 1.12, 95%CI 0.50 to 2.51; p=0.78).
Discussion
The primary aim of this study was to measure the seasonal variation in the prevalence of ED patients who meet the criteria for sepsis. The prevalence of ED patients who met the criteria for both “uncomplicated” sepsis and severe sepsis/septic shock, was higher in the winter month. We also found that the odds of a diagnosis of any type of sepsis or a diagnosis of severe sepsis/septic shock was 48% higher in the winter month, and 75% higher after adjusting for age and gender. Females were 28% more likely than males to receive a diagnosis of any type of sepsis or a diagnosis of severe sepsis/septic shock. The respiratory system was the most common source of sepsis during both study periods.
There is a paucity of studies investigating the seasonality of sepsis. Currently published studies focus on seasonal variation in patients hospitalised for sepsis3,7. To the best of our knowledge, this is the first study to measure the seasonality of sepsis in the ED setting. Our finding that the prevalence of ED patients who met the criteria for both “uncomplicated” sepsis and severe sepsis/septic shock is higher in the winter month is consistent with studies based on seasonal variation in hospital inpatients with sepsis3,7. Danai et al. reported that the seasonal incidence rate of sepsis increased 16.5% from a low of 41.4 in the autumn to a high of 48.6 cases per 100,000 in the winter3. Ore & Ireland reported that the average seasonal rate of sepsis with co- morbidities and complications increased 18.1% in winter compared to the summer7. The higher incidence and mortality of sepsis and severe sepsis in winter is related to respiratory sepsis, largely driven by respiratory tract infections8,9 and, possibly, the influence of photo-periodicity on leucocyte function10. Although, in general, males have a higher risk of developing sepsis than females, regardless of age11, in this study females were 28% more likely than males to meet the criteria for any type of sepsis. We hypothesise that the identified gender differences may be due to differences in the characteristics of the specific study population.
This study has some limitations4. Firstly, the systemic inflammatory response syndrome (SIRS) criteria used for sepsis diagnosis lacks specificity in the ED setting because infections account for only a quarter of adult SIRS patients12. Secondly, our case finding was based on clinical record documentation of a history suggestive of an infection. Therefore, any misdiagnoses by clinicians would alter the reported prevalence figures. However, by using this method we have enhanced the external validity of the study because it is reflective of actual clinical practice. The increased ED burden of sepsis in the winter demonstrated in this study can be used to inform resource allocation during the now annual winter hospital bed crisis.
Conflicts of Interest
None declared.
Funding Source: None
Correspondence
Dr. Abel Wakai, MD FRCEM, Emergency Care Research Unit (ECRU), Department of General Practice, Royal College of Surgeons in Ireland, 123 St. Stephen's Green, Dublin 2, Ireland
Email: [email protected]
Phone: +353 1 402 2305
Acknowledgements
The data presented here are as a result of a valuable collaborative process between the ED administrative, nursing, IT and medical staff in Beaumont Hospital. We thank them for their help and contribution.
References
1. National Clinical Effectiveness Committee (NCEC). Sepsis Management. National Clinical Guideline No. 6, November 2014 [cited 24 May 2017]. Available from: http://www.hse.ie/eng/about/Who/clinical/natclinprog/sepsis/sepsis%20management.pdf
2. McIntyre LA, Fergusson D, Cook DJ, Nair RC, Bell D, Dhingra V, Hutton B, Magder S, Hébert PC; Canadian Critical Care Trials Group. Resuscitating patients with early severe sepsis: a Canadian multicentre observational study. Can J Anaesth 2007;54(10):790-8.
3. Danai PA, Sinha S, Moss M, Haber MJ, Martin GS. Seasonal variation in the epidemiology of sepsis. Crit Care Med 2007;35(2):410-5.
4. McNevin C, McDowell, R., Fitzpatrick F, Gilligan P, Wakai A. What proportion of patients meet the criteria for uncomplicated sepsis in an Irish emergency department. Ir Med J 2016;109(7):435.
5. Health Service Executive (HSE). Sepsis. 2013. [Cited 24 May 2017] Available from: http://www.hse.ie/eng/about/Who/clinical/natclinprog/sepsis/Emergency%20Department%20Adult%20Sepsis%20Pathway.pdf
6. Clogg CC, Shihadeh SE. Statistical Models for Ordinal Variables. 1st ed. Thousand Oaks, California: SAGE Publications; 1994.
7. Ore T, Ireland P. Seasonal variability in sepsis hospitalisations. N Z Med J 2015;128(1417):61-2.
8. Dowell SF. Seasonal variation in host susceptibility and cycles of certain infectious diseases. Emerg Infect Dis 2001;7(3):369-74.
9. Griffin MR, Coffey CS, Neuzil KM, Mitchel EF, Jr., Wright PF, Edwards KM. Winter viruses: influenza- and respiratory syncytial virus-related morbidity in chronic lung disease. Arch Intern Med 2002;162(11):1229-36.
10. Yellon SM, Kim K, Hadley AR, Tran LT. Time course and role of the pineal gland in photoperiod control of innate immune cell functions in male Siberian hamsters. J Neuroimmunol 2005;161(1-2):137-44.
11. Angele MK, Pratschke S, Hubbard WJ, Chaudry IH. Gender differences in sepsis: cardiovascular and immunological aspects. Virulence 2014;5(1):12-9.
12. Horeczko T, Green JP, Panacek EA. Epidemiology of the systemic inflammatory response syndrome (SIRS) in the emergency department. West J of Emerg Med 2014;15(3):329-36
(P753)
