Severe Rhinitis Medicamentosa Successfully Treated with Rhinolight® Endonasal UV Phototherapy
P. Carson1, M. Lyons2
1. Slievemore Clinic, Dublin, Ireland
2. Edinburgh Medical School, Scotland
Abstract
Aim
Report successful application of UV endonasal phototherapy as a treatment for severe rhinitis medicamentosa and allergic rhinitis.
Methods
Allergic rhinitis confirmed by history and skin prick testing; rhinitis medicamentosa based on history. Both confirmed at nasendoscopy. Symptom score before & after treatment. Introduction of Rhinolight endonasal u/v phototherapy for allergic rhinitis. Single patient report.
Results
Successful remission of Rhinitis Medicamentosa confirmed with patient after eight sessions Rhinolight endonasal phototherapy. Use of nasal decongestant dropped from 2 bottles/daily x 4 years to zero. Symptoms reduced from 25 pre-treatment to 6 post-treatment.
Rhinitis medicamentosa is clinically characterized by nasal congestion without rhinorrhea, postnasal drip, or sneezing that begins after using a nasal decongestant for more than 3 days. Treatment involves discontinuation of the offending drug.
Discussion
Rhinolight endonasal phototherapy is a new treatment for allergic rhinitis and offered as last resort for a patient with untreated allergic rhinitis and overuse of topical decongestants. Patient reports a significant improvement in symptoms with cessation of topical decongestant.
Report a successful application of UV endonasal phototherapy as a treatment for severe rhinitis medicamentosa against a background of long standing allergic rhinitis.
Introduction
Rhinitis medicamentosa is intractable nasal congestion induced by overuse of medication, especially topical decongestants1. Treatment involves gradual weaning of topical decongestant, sometimes augmented by other anti-inflammatory or decongestant medications1.
Ultraviolet light phototherapy is a recognised treatment for allergic rhinitis2. Rhinolight® endonasal phototherapy is extensively in use in ENT/Allergy Centres in Europe, Japan and Australia. Dr PC has used Rhinolight to treat > 250 patients with intractable allergic rhinitis and shown it to provide substantial improvement in symptoms3.
Case Report
Mr. A is a 37 year old male with history of allergic rhinitis and asthma. He first attended in January 1995, aged 15 years. Skin prick testing showed positives to dust mite and pollens. Nasendoscopy revealed “severe nasal mucosal swelling with polypoid middle turbinates”. Treatment was short term Betamethasone drops BD; maintenance Fluticasone nasal spray 50 mcg BD and Chlorphenamine Meleate 4mg PRN. Asthma was managed with inhaled Beclazone 250mcg BD and Salbutamol 200mcg TDS.
The 1995 pollen season triggered severe hay fever requiring oral steroids and Azelastine hydrochloride nasal spray. He did not re-attend for follow up.
In January 2017 Mr. A re-presented with a permanently blocked nose. Working on radio and TV, he was using two bottles of Otrivine® per day for almost four years “so that I can breathe at night and sound normal on air”. Skin-prick testing confirmed dust mite and pollen sensitivities – same as in 1995. Nasendoscopy showed hyperaemic nasal mucosa with “blistering”, a finding most commonly seen allergic rhinitis.
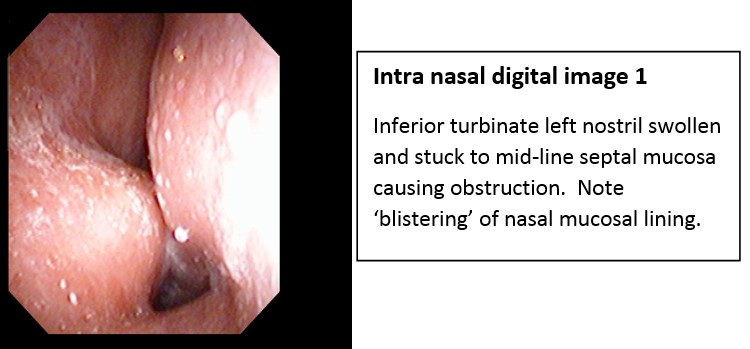
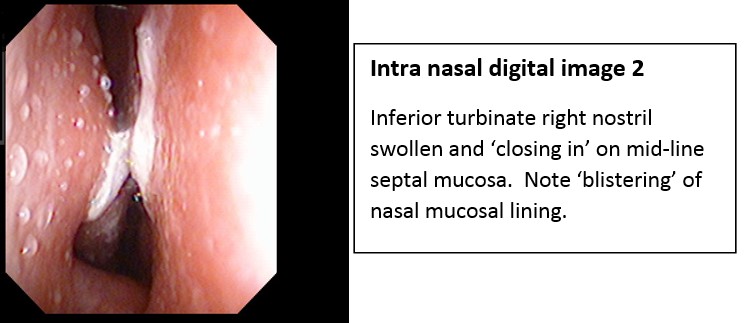
He refused standard anti-allergy therapies: “none of that has ever worked”.
He was offered Rhinolight® endonasal phototherapy to reverse the untreated allergic rhinitis: the background cause of nasal decongestant overuse and rhinitis medicamentosa. He received eight sessions of Rhinolight® –three over eight days and the subsequent five once a week. His pre-treatment symptom score (based on 0-10 of eight common symptoms in allergic rhinitis) was 25.
Mr. A failed to re-attend but in a recent e-mail exchange (02/03/2018) he stated he no-longer uses nasal decongestants, relying on Fluticasone nasal spray 50mcg 2 bd. His symptom score was 6 (rising to 9/10 during the pollen season), a significant reduction from the pre-treatment score of 25.
Discussion
The pathophysiology of rhinitis medicamentosa is unknown. It is histologically characterised by squamous cell metaplasia, epithelial oedema, goblet cell hyperplasia and inflammatory cell infiltration1. It is characterized by nasal congestion without rhinorrhoea, postnasal drip, or sneezing that begins after using a nasal decongestant for more than 3 days4.
While treatment relies on the discontinuation of the offending drug, some patients struggle with weaning. This may be due an underlying untreated allergic rhinitis.
This treatment was as last resort for a patient with excessive overuse of topical decongestants unwilling to attempt weaning. While post-treatment nasoendoscopy images are not available, the patient has reported a significant improvement in symptoms and has ceased topical decongestants.
We present a successful application of UV endonasal phototherapy as a treatment for severe rhinitis medicamentosa against a background of long standing allergic rhinitis.
Conflict of Interest
The author has no conflicts of interest to declare.
Correspondence Author
Paul Carson,
Slievemore Clinic,
Dublin,
Ireland
Email: [email protected]
References
1. Ramey JT, Bailen E, Lockey RF. Rhinitis medicamentosa. J Investig Allergol Clin Immunol. 2006;16(3):148–55.
2. Kemény L, Koreck A. Ultraviolet light phototherapy for allergic rhinitis. J Photochem Photobiol B Biol. 2007 Apr;87(1):58–65.
3. Carson P, Lyons M. O.017 Rhinolight® endonasal phototherapy in primary care: First impressions - Presented at the 2017 Annual Meeting of the British Society for Allergy and Clinical Immunology. Clin Exp Allergy. 2017 Dec;47(12):1678–721.
4. Dykewicz MS, Fineman S, Skoner DP, Nicklas R, Lee R, Blessing-Moore J, et al. Diagnosis and management of rhinitis: complete guidelines of the Joint Task Force on Practice Parameters in Allergy, Asthma and Immunology. American Academy of Allergy, Asthma, and Immunology. Ann Allergy Asthma Immunol. 1998 Nov;81(5 Pt 2):478–518.
P874
