Botulinum Toxin in the Management of Sialorrhoea in Acquired Brain Injury
A Carroll, B McGlone
National Rehabilitation Hospital, Rochestown Ave, Dun Laoghaire, Co Dublin
Abstract
Sialorrhoea as a consequence of severe acquired brain injury can significantly negatively impact on quality of life. Medications used in its management have many side effects which can cause problems in the severely disabled. Botulinum toxin is an effective treatment of sialorrhoea in a number of neurological conditions but may also have a role to play in the management of sialorrhoea following severe ABI. We report on 4 cases of sialorrhoea following acquired brain injury causing a variety of problems, whose parotid glands were injected with Botulinum toxin type A (Dysport) 50mu each, under ultrasound guidance. All cases had a clinically and statistically significant reduction in drooling as measured by the teacher drooling scale (p=0.005) and carers Visual Analogue Scale (p=0.012). There were no side effects reported. Botulinum toxin is an effective treatment for sialorrhoea associated with acquired brain injury.
Introduction
Sialorrhoea is a problem in a variety of neurological disorders and can result in physical and psychosocial consequences for healthcare providers, patients and families. Successful management can contribute to improved biopsychosocial outcomes1. Saliva has a number of important functions that are important to consider when considering treatment options. There are a variety of treatment options, but none are universally effective and many have significant complications. Anticholinergic medication, whilst effective has disabling side effects. Botulinum toxin is a local effective treatment for sialorrhoea with minimal side effects2-10 but its use in the treatment of sialorrhoea in patients with ABI has not been extensively studied. We report on 4 cases with ABI and sialorrhoea treated with Botulinum toxin.
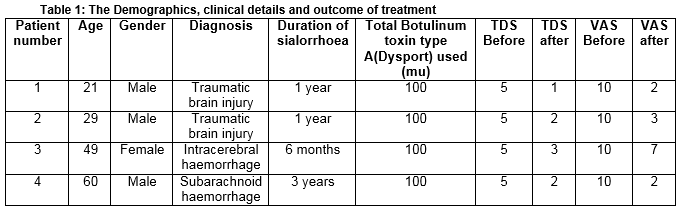
Case Report
Retrospective analysis of in-patient case notes of four adult patients with ABI and sialorrhoea who were treated by injection of Botulinum toxin type A (Dysport) 50mu into the parotid glands under ultrasound guidance. All patients had been assessed by the MDT, had appropriate seating systems and optimal positioning and had been unsuccessfully treated with anticholinergic agents. The parotid glands had been imaged using high-resolution ultrasound. Botulinum toxin type A (Dysport) 50 mu was reconstituted with 0.9% sodium chloride solution to 100 mu/ml and 50mu was injected into each gland. The glands were re-imaged post injection. The recorded subjective and objective measurement of the severity of drooling before and after botulinum toxin A injections (Teacher drooling scale [TDS] and a visual analogue scale [VAS] for carers to report on effect of treatment) were documented. The VAS scale used (before and after injection) was a 100 mm horizontal line marked from 0 (normal salivation) up to 100 (worst possible drooling). The Demographics, clinical details and outcome of treatment are shown in table 1. There were no side effects reported. SpSS for Windows was used for statistical analysis with statistical significance determined at p= 0.05 and the results were analysed using paired t-tests. The change in Teacher drooling was statistically significant (p=0.005) [SD = 0.816] as was change in VAS (p=0.012) [SD = 2.38]. The results were also clinically significant with a marked reduction in severity of sialorrhoea as shown in table 1. This effect was maintained at 6 months.
Discussion
This case series shows the beneficial effect of intraglandular injection of botulinum toxin in the management of sialorrhoea associated with ABI with no side effects. Ultrasound guidance minimises side effects and ensures accurate intraglandular injection and avoids extracapsular migration. Further RCTs are required to fully evaluate this new modality of treatment.
Conflict of Interest:
The authors declare that they do not have financial or personal relationships with other people or organisations that could inappropriately influence or bias the content in this article in order for self-gratification or undue gains.
Correspondence: Aine Carroll
National Rehabilitation Hospital, Rochestown Ave, Dun Laoghaire, Co Dublin
Email: [email protected]
References
1. Bavikatte, G. Poh L. S, Hassoon A. Management of Drooling of saliva. BJMP 2012;5:a507
2. Porta M, Gamba M, Bertacchi G, Vaj P. Treatment of sialorrhoea with ultrasound guided botulinum toxin type A injection in patients with neurological disorders. J Neurol Neurosurg Psychiatry 2000; 70:583-540.
3. Ultrasound-guided versus ‘blind’ intraparotid injections of botulinum toxin-A for the treatment of sialorrhoea in patients with Parkinson’s disease 2004;106:93-96
4. Lim M, Mace A, Reza Nouraei SA, Sandhu G. Botulinum toxin in the management of sialorrhoea: a systematic review. Clinical Otolaryngology 2006; 31: 267–272.
5. Truong D.D., Bhidayasiri, R., Evidence for the effectiveness of botulinum toxin for sialorrhoea. Journal of Neural Transmission 2008;115: 631-635
6. Heinen F. Sialorrhoea in paediatric neurology - the long way from case series to clinical studies. Neuropediatrics. 2008 Aug; 39:195.
7. Squires N1, Humberstone M, Wills A, Arthur A The use of botulinum toxin injections to manage drooling in amyotrophic lateral sclerosis/motor neurone disease: a systematic review.Dysphagia. 2014 Aug;29:500
8. Rodwell K1, Edwards P, Ware RS, Boyd R. Salivary gland botulinum toxin injections for drooling in children with cerebral palsy and neurodevelopmental disability: a systematic review. Dev Med Child Neurol. 2012 Nov;54:977-87.
9. Wilken B, Aslami B, Backes H. Successful treatment of drooling in children with neurological disorders with botulinum toxin A or B. Neuropediatrics. 2008 Aug; 39:200-4.
10. Stone CA, O'Leary N. Systematic review of the effectiveness of botulinum toxin or radiotherapy for sialorrhoea in patients with amyotrophic lateral sclerosis. J Pain Symptom Manage. 2009 Feb; 37:246-58.
p425