Can Early Changes in Vital signs Predict Duration of Antibiotic Therapy in Suspected Neonatal Sepsis?
M. McGovern1,2, P. Morrissey1,2, E. Ryan1,2
1. Department of Paediatrics and Neonatology University College Hospital Galway
2. Academic Department of Paediatrics, National University of Ireland, Galway
Abstract
Aims
Suspected sepsis remains a leading causes of Neonatal Intensive Care Unit admission, with infants often receiving 48-72 hours of empirical antibiotic therapy. Early in treatment it is difficult to predict infants who will require prolonged antibiotic therapy. Our aim was to assess if vital sign measurements in the initial period of treatment can predict those neonates requiring prolonged antibiotic therapy in term and late-preterm infants.
Methods
Data was retrospectively collected over 1 year on neonates admitted to our institute for antibiotics. Infants were classified as standard (<48hours duration) or prolonged (>48hours duration) antibiotic therapy.
Results
Respiratory rate on admission and 12 hours after initiation of therapy correlated significantly with duration of antibiotic therapy and infants requiring prolonged therapy were more likely to have one or more abnormal vital signs 12 hours after initiation of treatment (p<0.05).
Conclusion
Respiratory rate shows a weak positive correlation with antibiotic duration. Infants requiring prolonged therapy were more likely to have abnormal vital signs 12 hours after initiating antibiotic therapy. Changes in vital signs maybe useful in detecting infants who will require prolonged antibiotic therapy.
Introduction
Neonatal sepsis has traditionally been a major contributor to neonatal morbidity and mortality.1,2 As a result, paediatricians are always aware of the possibility of sepsis as a cause of any change in clinical parameters in the neonatal period. Due to the high morbidity associated with delay in treatment, it is common practice to perform sepsis evaluation and empirical antibiotic therapy for infants in whom there is any clinical suspicion. While policies vary between centres it is commonplace for such infants to have blood drawn for complete blood count (CBC), C-Reactive Protein (CRP) and blood culture with further testing depending on clinical assessment and local guidance.3 Infants >35 weeks are commonly admitted from postnatal ward for this reason and often remain in the Neonatal Intensive Care Unit (NICU) to allow close monitoring and administration of empirical antibiotic therapy. Duration and choice of antibiotic therapy differs between centres,4 with many opting to continue antibiotic therapy empirically until initial blood cultures are confirmed to be negative and the infant’s clinical examination is unremarkable. In our centre, a secondary level neonatal unit with 2974 births during the year of study, it is our policy to provide 48hours empirical intravenous antibiotic therapy and if blood cultures are negative and the clinical scenario is reassuring treatment is discontinued. It is not uncommon however, for some infants to require longer duration of therapy depending on the results of initial laboratory testing or persisting clinical concerns on the part of the treating paediatrician. Predicting which infants will require prolonged antibiotic therapy would be of great benefit in terms of counselling parents, laboratory investigation and planning service delivery. We designed this retrospective cohort study to assess if alterations in vital parameters in the initial period of admission to the NICU can predict those infants who subsequently require prolonged antibiotic therapy in term and late-preterm neonates.
Methodology
We reviewed our computerised admission records over a one-year period (Jan 2015-Dec 2015) to identify those infants >35 weeks gestation who were admitted to the NICU for antibiotic therapy. Infants were divided into two groups based on duration of antibiotic therapy: those receiving standard 48hours of empirical intravenous antibiotic therapy (classified as short duration), and those treated with prolonged courses of >48hours duration. Inclusion criteria were as follows: >35 weeks gestation, admitted due to suspected sepsis. Exclusion criteria were as follows: <35weeks gestation, congenital abnormality, external referrals from other centres and severe neonatal hypoxia. For each infant we collected the following information: gestation, birthweight, sex, maternal risk factors for sepsis, delivery and resuscitation details, age in hours upon admission, results of sepsis evaluation, vital signs recorded on admission, vital signs recorded at 12 hours after admission and duration of antibiotic therapy. Vital signs recorded in each case were: respiratory rate, heart rate, oxygen saturations, blood pressure and temperature. Paired t-testing and independent sample t-testing were used to compare means, Pearson's correlation coefficient was used to assess for relationships between predictor and outcome variables and x2 or Fisher’s exact tests were used to compare categorical data. Statistical analysis was performed on Minitab 17. Ethical approval was obtained from the local Research Ethics Committee.
Results
Ninety-six infants >35 weeks gestation were identified as being eligible for inclusion; thirty-one female, sixty-five male. Mean gestation on admission was 39.33 weeks, mean birthweight on admission was 3.5kg and mean age on admission was 6.25 hours. Eighty had standard antibiotic regimens of 48 hours duration, sixteen had prolonged antibiotic therapy lasting >48 hours (mean duration 129hours). Table 1 compares the maternal sepsis risk factors present in each case. Gestation, birth weight, age on admission, white cell count and neutrophil count on admission did not differ significantly between groups. CRP was significantly higher in the group with prolonged therapy (1.7 vs. 16.7, p= 0.038, 95% CI: 29.05; 0.96).
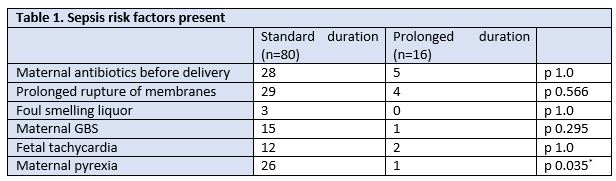
No significant differences existed between groups in the need for respiratory support on admission (p>0.05). After twelve hours of treatment the number of vital signs outside normal limits had declined significantly in those who had short-course antibiotic therapy (p=0.00, 95% CI: 0.396; 0.809), but not in those who subsequently required prolonged antibiotic therapy (p=0.791, 95% CI: -0.432; 0.557). Those with prolonged antibiotic therapy were more likely to have vital signs outside normal limits at 12 hours (p=0.012, Likelihood Ratio Chi-Square = 6.259). Analysis showed weak but significant correlations between the number of days of antibiotic therapy and each of the following: respiratory rate on admission (r=0.203, p=0.047), respiratory rate at 12 hours after admission (r=0.309, p=0.003) and the number of deranged vital signs at 12hours (r=0.241, p=0.018).
Discussion
Neonatal sepsis remains a key area of interest within paediatrics and research to date has yielded improvements in sepsis rates and mortality in the newborn period.2Much of this improvement is attributable to high-quality care in the antenatal and early postpartum period, with particular success seen in the prevention of Group B Streptococcus sepsis through the use of intrapartum antibiotics.5Despite this, sepsis remains a leading cause of neonatal morbidity and mortality, 1, 2and there remains great variation in practice between centres in investigation and treatment of neonates with suspected sepsis. It is notable that there is an increasing trend towards 36 hours of empirical antibiotic therapy instead of the traditional 48 hours among term infants with suspected sepsis in many neonatal centres. This is supported by recent NICE guidelines which recommend cessation of treatment if clinical picture and laboratory parameters are reassuring.6 Many centres, such as our own, opt to perform a basic blood work-up (CBC, CRP, blood culture) and pending these results and clinical course of the infant more testing may be ordered. While laboratory techniques for the detection of neonatal infection have utility in diagnosis, they lack specificity and sensitivity early in the course of disease.7Because of the limited utility of laboratory investigation early in the course of neonatal sepsis, paediatricians often rely on the clinical status of the infant to inform decision-making, with published data confirming laboratory results are rarely informative before clinical symptoms in infants >35 weeks at risk of sepsis.8Significant variability still exists between units in terms of antimicrobial prescription in neonates with suspected sepsis.4Although expert guidance has been provided in the area,3there remains significant debate on the correct approach to investigation and treatment of infants admitted to the NICU with suspected sepsis, and this is especially so with regard to the duration of antimicrobial therapy in infants who have negative blood cultures.9A recent review by Cotton et al. reflected this, concluding that there is insufficient evidence to suggest a standardised approach to this common clinical scenario,10and hence it is often an individualised decision based on laboratory markers, clinical examination and clinician experience.
While infants with suspected sepsis who remain clinically unwell after several days are likely to require prolonged treatment, there is very little data on how these infants can be clinically identified at an early stage. Our findings suggest that respiratory rate may be a useful clinical parameter early in admission for predicting those infants who will require prolonged admission and treatment, as a weak correlation was demonstrated with the duration of therapy both on admission to the NICU and 12 hours later. The number of vital signs outside normal limits at 12 hours may also be a useful measure of those infants who will require a longer course of antibiotic therapy. However, since this measure was most often due to persistently abnormal oxygen saturations or respiration rate, this may simply be representative of abnormal respiratory status in these infants. Our findings are in keeping with previously published work suggesting that respiratory dysfunction is the commonest presentation of sepsis in the preterm population 11 and respiratory signs such as apnoea and tachypnoea are among the best predictors of blood culture positivity in neonates with suspected sepsis 12. Tachypnoea is a common neonatal presentation and not specific to sepsis and it has recently been suggested that non-conventional vital sign monitoring such as heart rate variability may be better predictors 13, 14 than traditional vital signs such as those recorded in this study.
Our study found that sepsis risks in the mother were broadly similar between groups, apart from maternal pyrexia which was more frequent in those requiring standard duration therapy. Likewise, the basic demographic information was comparable between groups representing infants of similar weight, age on admission and gestation. CRP was found to be higher in the group requiring prolonged therapy.
In terms of limitations we acknowledge that our study was retrospective and therefore is limited in scope, however this methodology had the advantage of not influencing clinician decisions on management. We also acknowledge that the sample size in our study is limited with only 96 patients overall and only 16 of these receiving prolonged antibiotic therapy, however all patients were consecutive presentations and represented all potentially eligible infants in the period of study.
Subtle clinical signs are present early in the course of treatment which may help to predict those infants who will require prolonged NICU admission and antibiotic therapy. Respiratory rate both on admission and 12 hours after the initiation of therapy showed weak but significant correlations with the duration of antibiotic therapy, and those requiring prolonged therapy were more likely to have abnormal vital signs recorded 12 hours after admission. Due to the weak associations demonstrated, at present the findings of this study cannot be incorporated directly into clinical practice. However, changes in vital parameters over time should be investigated prospectively in a larger cohort as they may represent useful predictors of clinical course in infants >35 weeks admitted to the NICU with suspected sepsis.
Conflict of Interest:
The authors declare that they have no conflict of interest.
Correspondence Author:
Dr. Matthew McGovern
Email: [email protected]
Acknowledgements:
The authors would like to thank the staff in the Neonatal department in Galway University Hospital for their assistance with enrolment and the medical records department in our institute for obtaining records on participating patients.
References
1. Camacho-Gonzalez A, Spearman PW, Stoll BJ. Neonatal infectious diseases: evaluation of neonatal sepsis. Pediatric clinics of North America. 2013;60(2):367-89.
2. Shane AL, Stoll BJ. Neonatal sepsis: progress towards improved outcomes. The Journal of infection. 2014;68 Suppl 1:S24-32.
3. Polin RA. Management of neonates with suspected or proven early-onset bacterial sepsis. Pediatrics. 2012;129(5):1006-15.
4. Oliver EA, Reagan PB, Slaughter JL, Buhimschi CS, Buhimschi IA. Patterns of Empiric Antibiotic Administration for Presumed Early-Onset Neonatal Sepsis in Neonatal Intensive Care Units in the United States. American journal of perinatology. 2017;34(7):640-7.
5. Verani JR, McGee L, Schrag SJ. Prevention of perinatal group B streptococcal disease--revised guidelines from CDC, 2010. MMWR Recommendations and reports : Morbidity and mortality weekly report Recommendations and reports. 2010;59(Rr-10):1-36.
6. NICE. Neonatal infection (early onset): antibiotics for prevention and treatment 2017 [updated 2017; cited 2019. Available from: https://www.nice.org.uk/guidance/cg149/chapter/1-guidance.
7. Benitz WE. Adjunct laboratory tests in the diagnosis of early-onset neonatal sepsis. Clinics in perinatology. 2010;37(2):421-38.
8. Berardi A, Fornaciari S, Rossi C, Patianna V, Bacchi Reggiani ML, Ferrari F, Neri I, Ferrari F. Safety of physical examination alone for managing well-appearing neonates >/= 35 weeks' gestation at risk for early-onset sepsis. The journal of maternal-fetal & neonatal medicine : the official journal of the European Association of Perinatal Medicine, the Federation of Asia and Oceania Perinatal Societies, the International Society of Perinatal Obstet. 2015;28(10):1123-7.
9. Cotten CM, Benjamin DK, Jr., Smith PB, Stoll BJ, Spitzer AR, Clark RH. Empirical antibiotic therapy for suspected early-onset bacterial sepsis. Pediatrics. 2012;130(4):e1052-3; author reply e5-7.
10. Cotten CM, Smith PB. Duration of empirical antibiotic therapy for infants suspected of early-onset sepsis. Current opinion in pediatrics. 2013;25(2):167-71.
11. Gonzalez BE, Mercado CK, Johnson L, Brodsky NL, Bhandari V. Early markers of late-onset sepsis in premature neonates: clinical, hematological and cytokine profile. Journal of perinatal medicine. 2003;31(1):60-8.
12. Ohlin A, Björkqvist M, Montgomery SM, Schollin J. Clinical signs and CRP values associated with blood culture results in neonates evaluated for suspected sepsis. Acta Paediatrica. 2010;99(11):1635-40.
13. Griffin MP, Lake DE, O'Shea TM, Moorman JR. Heart rate characteristics and clinical signs in neonatal sepsis. Pediatric research. 2007;61(2):222-7.
14. Bohanon FJ, Mrazek AA, Shabana MT, Mims S, Radhakrishnan GL, Kramer GC, Radhakrishnan RS. Heart rate variability analysis is more sensitive at identifying neonatal sepsis than conventional vital signs. American journal of surgery. 2015;210(4):661-7.
P909
