Transcatheter Mitral Valve-in-Ring Implantation
RE Tanner1, J McCarthy2, K P Walsh1, G Blake1, I P Casserly1
1Department of Cardiology, Mater Misericordiae University Hospital.
2 Department of Cardiothoracic Surgery, Mater Misericordiae University Hospital.
Abstract
Failed surgical mitral valve repair using an annuloplasty ring has traditionally been treated with surgical valve replacement or repair1. For patients at high risk for repeat open heart surgery, placement of a trans-catheter aortic valve (i.e., TAVI valve) within the mitral ring (i.e., Mitral-Valve-in-Ring, MViR) has emerged as a novel alternative treatment strategy2-5 . We describe our experience of a failed mitral valve repair that was successfully treated with a TAVI valve delivered via the trans-septal approach, and summarise the data relating to this emerging treatment strategy.
Case report
A 56-year-old lady underwent emergent mitral valve repair in August 2012 for treatment of acute severe mitral regurgitation (MR) secondary to myxomatous mitral valve disease. A 30mm Carpentier-Edwards Physio II mitral annuloplasty ring (Edwards Lifescience, CA) was used. Co-morbidities included atrial fibrillation, raised body mass index of 38, bicuspid aortic valve with mild stenosis and stable thoracic aortic aneurysm (4.5cm). She represented approximately three years later with exertional shortness of breath (NYHA class III). A transoesophageal echo (TOE) showed severe valvular MR and an ejection fraction (EF) of 55%. Right heart catheterisation documented severe pulmonary hypertension (pulmonary artery systolic pressure 97mmHg, mean pulmonary pressure 60mmHg). Following discussion of the heart team and discussion with the patient, it was decided to attempt trans-septal delivery of a TAVI valve within the mitral ring, with the understanding that she will likely need a surgery within the next decade to deal with her bicuspid aortic valve disease (currently causing stenosis of mild severity) which could be combined with mitral valve surgery if needed. The severity of her pulmonary hypertension was also felt to favour a percutaneous approach.
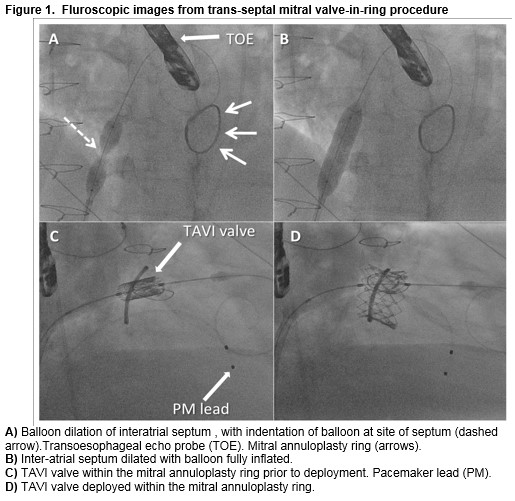
Under general anaesthetic, the right common femoral vein (CFV) was accessed and an 8Fr sheath was placed. With transoesophageal echo (TOE) guidance, a high posterior trans-septal puncture was performed which facilitated delivery of a large curve Safari wire (Boston Scientific) into the left atrium. The inter-atrial septum was then dilated with a 12mm x 4cm Armada 35 balloon (Abbott Vascular) (Figure 1a-b). A Swan Ganz catheter with the balloon inflated was then delivered across the mitral valve allowing delivery of the large curve Safari guide into the left ventricle. Under rapid pacing of the right ventricle using a temporary pacemaker lead introduced from the left CFV, a 29mm Edwards Sapien XT TAVI valve was deployed within the mitral valve ring (Figure 1c-d).
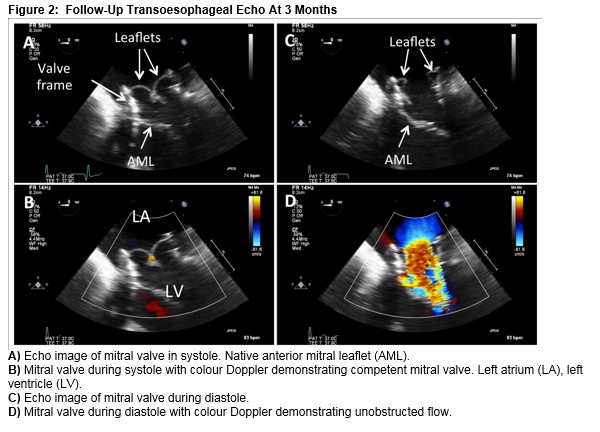
The patient made an excellent recovery and was discharged home on day two post-procedure. A TOE at three months showed a well seated TAVI valve within the mitral annuloplasty ring with a trivial jet of paravalvular MR (Figure 2a-d).
Discussion
This case highlights the effectiveness of the strategy of using a TAVI valve introduced via the trans-septal route to treat a failed mitral valve repair surgery that used an annuloplasty ring. To our knowledge this is the first time this procedure has been performed in Ireland. This MViR treatment option has only emerged in the last 4-5 years with the evolution of TAVI valve technology and clinical experience.
Our experience with this case and a similar case that used the alternate trans-apical approach was that the technique was remarkably straightforward and the procedures were very well tolerated. In the trans-apical case a mini-thoractomy performed at the site of the left ventricular apex allowed access for a delivery sheath [23Fr Ascendrasheath (Edwards Lifescience)] to be directed from the LV into the left atrium across the mitral valve ring over a super stiff Amplatz wire. A 29mm Edwards Sapien XT valve was then delivered through the 23Fr sheath.
Device delivery across the septum following balloon dilatation of the inter-atrial septum was uncomplicated and positioning of the valve across the mitral annuloplaty ring was only marginally more challenging with this approach compared to the trans-apical approach. We would certainly favour the trans-septal approach in the future, driven largely by the desire to eliminate the morbidity associated with trans-apical exposure.
Patients with a complete, semi-compliant mitral annuloplasty ring as was used in this case are likely the ideal candidates for this MViR strategy. The complete ring provides for greater device stability and the semi-compliant nature of the ring allows for circularisation of the ring folowing deployment of the valve, which minimizes the risk of paravalular leak.
The majority of the 65 published MViR cases have been treated using the Edwards Sapien family of balloon-expandable TAVI valves with delivery via the trans-apical approach. Among these 65 cases, complications included; left ventricular outflow tract obstruction (n=4, 6%), TAVI valve migration (n=2, 3%), and ring dehiscence (n=2, 3%). Five (8%) deaths within 30 days of the procedure were reported2,4-7. An international valve-in-valve registry reported a higher 30-day mortality (13%) for 88 patients undergoing MViR procedures8. This data highlights that MViR should be offered at specialised heart valve centres where multidisciplinary teams work together to try to maximize clinical outcomes in this complex patient group.
Conflicts of Interest
The authors have no conflicts of interest to declare
Corresponding Author
Prof Ivan P Casserly, Mater Misericordiae University Hospital, Eccles St, Dublin 7.
E-mail: [email protected]
References
1. Dumont E, Gillinov AM, Blackstone EH, Sabik JF, 3rd, Svensson LG, Mihaljevic T, Houghtaling PL, Lytle BW. Reoperation after mitral valve repair for degenerative disease. Ann Thorac Surg. 2007 Aug;84(2):444-50; discussion 50.
2. Eleid MF, Cabalka AK, Williams MR, Whisenant BK, Alli OO, Fam N, Pollack, PM, Barrow F, Malouf JF, Nishimura RA, Joyce LD, Dearani JA, Rihal CS. Percutaneous Transvenous Transseptal Transcatheter Valve Implantation in Failed Bioprosthetic Mitral Valves, Ring Annuloplasty, and Severe Mitral Annular Calcification. JACC Cardiovasc Interv. 2016 Apr 11.
3. Attizzani GF, Cheung Tam C, Markowitz A. Transcatheter mitral valve-in-ring implantation in prohibitive surgical risk patients: Single center initial experience in the United States. Catheterization and Cardiovascular Interventions. 2016 Dec;88(7):E233-E238
4. Descoutures F, Himbert D, Maisano F, Casselman F, de Weger A, Bodea O, Van der Kley F, Colombo A, Giannini C, Rein KA, De Bruyne B, Dahle G, Alfieri O, Vahanian A. Transcatheter valve-in-ring implantation after failure of surgical mitral repair. European Journal of Cardio-Thoracic Surgery. 2013;44(1):e8-e15.
5. Latib A, Ruparelia N, Bijuklic K, De Marco F, Gatto F, Hansen L, Oxbek C, Greilach, Peter, Bruschi G, Rieb FC, Alfieri O, Colombo A, Schofer J. First-in-man transcatheter mitral valve-in-ring implantation with a repositionable and retrievable aortic valve prosthesis. EuroIntervention. 2016;11(10):1148-52
6. Van Rosendael P, van der Kley F, Martina B, Palmen M, Delgado V, Schillinger W, Von Bardeleben RS, Yeo KK , Ho KW, Jack Tan WC. How should I treat recurrent concomitant para-ring and valvular mitral regurgitation after surgical mitral valve repair in a high-risk patient? EuroIntervention. 2015;10(12):1488-92
7. Lauterbach M, Sontag B, Paraforos A, Friedrich I, Hauptmann K-E. Transcatheter valve-in-ring implantation of a repositionable valve system for treatment of severe mitral regurgitation. Catheterization and Cardiovascular Interventions. 2016 Nov 15;88(6):E183-E190
8. Divir D. Transcatheter mitral valve-in-valve implantation advantageous in failed bioprosthetic valves. European Association of Percutaneous Cardiovascular Interventions; July; Paris 2015. URL: http://www.ecardiologynews.com/specialty-focus/interventional-cardiology-surgery/single-article-page/transcatheter-mitral-valve-in-valve-implantation-advantageous-in-failed-bioprosthetic-valves/4a421da670a1760bb12625785bca7a36.html
(P758)
